CV Examples for UK Jobs and Industries
See professional CV examples for a wide range of popular UK professions, all created by our certified career experts. Browse through job-winning CV samples, get inspired, and have a ready-to-use, professional CV in just minutes!

Our customers have been hired by*:
Most popular CV examples
See our 12 most popular CV samples for various careers and experience levels. Choosing one of our great CV examples will help you get started.
Job-specific CV examples
Browse our library of CV examples listed by job title and categorised by industry.
Commerce CV examples
Want to land a deal of your life? Read and personalise these professional CV examples for retail and sales jobs using expert tips from each article.
Creative careers CV examples
It takes hard work to translate artistry into a job application. Get inspired and follow the instructions with these creative CV samples to craft your CV.
Education CV examples
These CV examples for UK-based educators will quickly teach you how to present your qualifications and easily pass recruitment. Use our expert advice to personalise a CV sample that matches your career profile.
Supply teacher CV

Teacher CV

SEN teaching assistant CV

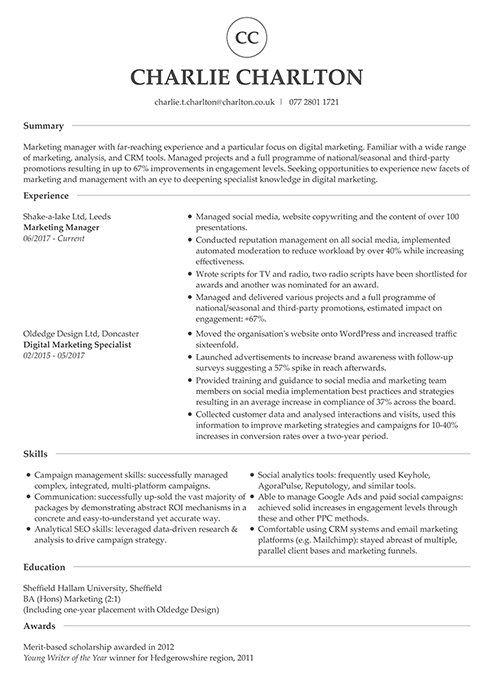
Finance CV examples
You speak numbers, but hiring managers expect words. These UK CV examples can help you translate KPIs into a successful job application for accounting, finance, or business sectors.
Food and drink CV examples
Have a look at these sweet CV samples and learn how to present your professional skills and experience, to cook up a CV that appeals to recruiters.
Healthcare CV examples
Whether you're applying in medicine or mental health, these CV samples demonstrate how to combine proven skill sets with a thoughtful, human approach to care.
Hairdresser CV

Assistant psychologist CV

Care assistant CV

Legal CV examples
If you're looking for good CV examples for legal professionals, you're in the right place. Each guide features invaluable tips from career experts to help you make a strong case with your CV.
Office jobs CV examples
Applying for an office or administrative job in the UK? Our CV samples will show you how to describe relevant experience and prove you've got the transferable skills needed to handle daily administrative duties.
HR manager CV

Administrator CV

Customer service CV

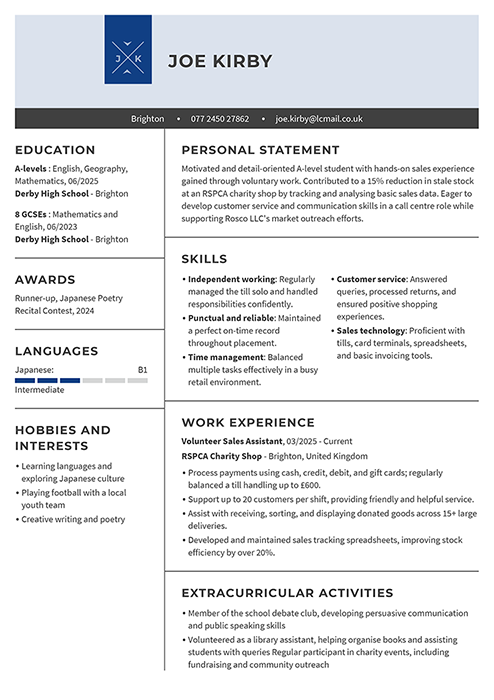
Student CV examples
See student CV examples that teach you how entry-level applicants can present relevant experience. These sample CVs will show you how to use educational achievements to your benefit.
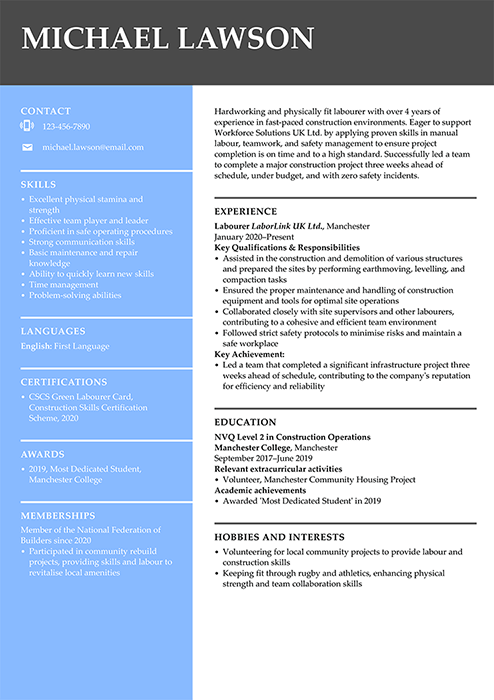
Technical CV examples
These CV examples for technical and industry jobs combine hard skills and interpersonal qualities needed for any job. Follow our expert guides to customise the samples with your personal information.
Technology CV examples
A successful CV for a tech-based job needs more than computer skills. Check these professional CV samples to learn about what to include in a CV for tech roles to impress the hiring managers.
Computer scientist CV
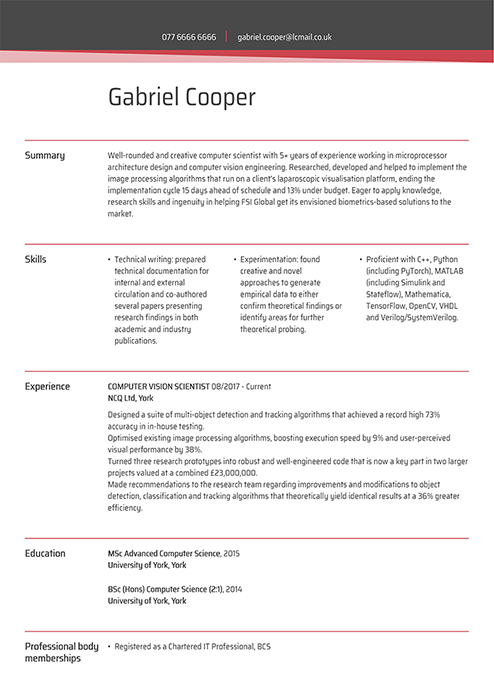
Cyber security CV

DevOps engineer CV

Other CV examples
Discover CV examples for various situations and jobs that don't belong in other categories. Follow the advice featured in each article to learn how to adapt each CV sample to your specific situation.
CV samples you can copy, adjust, and use
Writing a CV doesn't have to be a hassle. Using a good CV example can save you a lot of time and make your life easier.
All of our CV examples are written by career professionals, so you can be sure they meet modern recruitment standards. That means you don't have to worry about technicalities, and you can focus on what's important-presenting yourself in the best possible way.
What's more, each example of a CV comes with a dedicated guide where we share tips and tricks on how to make the most of your CV.

See what real users say about LiveCareer
LiveCareer customers got the job on average 48 days faster.*
*based on a survey of 258 LiveCareer users
How to create a CV based on examples?
First of all, don’t do mindless copypasta. Our CV examples have been carefully crafted by certified CV writers but, at the end of the day, they’re examples. You should use them as a point of reference while tailoring every CV you send—it’s the single most effective job-hunting strategy.
Make sure your experience, skills, and personal profile match the requirements from the job ad. Also, make sure to add the right keywords. You guessed it, the ones that are used in the job ad. If you need help, check our comprehensive guide on how to write a CV.
Our CV maker comes with ready-made content for every CV section, so with little adjustments and tweaks, you can have a job-winning CV ready in minutes.

CV sample anatomy
No matter your needs and the exact CV format you go for, your CV has to include some mandatory sections—those that list the key information recruiters are after.
6 parts every good CV should have:
- Contact information: Give your personal details, mainly your full name, email address, phone number, and LinkedIn profile. A physical address is not required. If you’re professionally active on social platforms such as Twitter or GitHub, include those links too.
- Personal profile: Provide a brief summary of your competencies, career achievements, and an offer of what you can do for the employer.
- Work history: List of your past employers, in reverse-chronological order. In each entry, include the company name, your job title, and 3–6 bullet points summarising your responsibilities and accomplishments.
- Education section: Add the highest degree of education. List the name of the institution and your degree. If your past degrees are highly relevant, you can add them in a reverse-chronological order. Provide relevant coursework, honours, etc., if you have little work experience.
- Job skills: the most relevant skills that prove you’ll be able to excel in the job. Limit yourself to 10 skills. Remember to include both hard and soft skills. Add an achievement to each skill if you want to provide additional qualifications.
- Additional sections: anything that proves your worth and validates your skill set. Certifications, additional training, conferences, publications, or volunteer work are popular choices.
How to use a CV sample?
- 1
In the CV creator, answer how much guidance you need when writing a CV, and then, pick a CV template you like the most. Follow our guidance and utilise AI-generated inputs to fill out every section of your CV with professional and tailored information.
- 2
When writing a CV with no experience, focus on your skills and education. If you’re an experienced candidate, apply special care to your work history section. Include 1–3 bonus sections to stand out as a more versatile and dedicated candidate.
- 3
Remember that, according to the UK standards, a good CV length is up to 2 A4 pages. The shorter you can make it, the better. Also, in most situations, opt for a reverse-chronological CV format.
- 4
The CV layout, content, and vocabulary may differ depending on the country. If you’re applying in a different country than the UK, remember to familiarise yourself with the recruitment rules prevailing there.
- 5
Before sending your CV, check for any typical spelling or punctuation errors. Our CV builder has an integrated spell-checker to help you identify mistakes.
- 6
Send your CV in a PDF file. This way, your document will retain its original CV format, and the recruiter will correctly display it on any device. Use a different file type only when it’s stated in the job posting.
Take the first step towards career success
Build a professional CV with LiveCareer’s intuitive CV builder. Simple, fast, and tailored to your goals. Get started today!
Build my CV
Frequently asked questions about CV examples
Yes, the CV examples and all other resources available on our blog are free. Our experts left plenty of actionable advice on whether you want to learn how to create a CV from scratch or improve the one you already have, with tips for putting skills on your CV and more.
Every CV example is different, as the content highly depends on the job it's created for. But they all have a CV sample you can copy and adjust to your experience, tips for every CV section with examples put into practice, and a list of other CV examples articles similar enough to get even more inspired.
There are two ways. The first is to use our builder, which will format the template for you and give you ready content suggestions for all sections based on your personal information. The other is to find a CV sample in our library, prepare a rough template in a text editor, and personalise it, taking advice from our LiveCareer UK career experts.
Research is very important to our writers, who follow hiring and recruitment trends in all of their articles. Recently, they reviewed data from an internal study using 6 million CVs created in our builder and updated their advice with the findings, which will now help you make even more informed decisions.
Yes, we do! Just like our CV examples, our cover letter samples are completely free and come with expert tips for every paragraph. Read our dedicated cover letter writing guide, and then hop into our cover letter builder to supplement your CV with a strong letter.